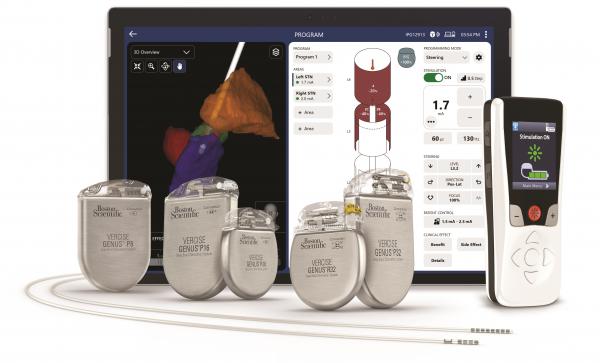
MARLBOROUGH, Mass., Jul. 11, 2023 -- Boston Scientific Corporation (NYSE: BSX) today announced it has received U.S. Food and Drug Administration (FDA) approval for the Vercise™ Neural Navigator 5 Software, which when used as part of the Vercise Genus™ Deep Brain Stimulation (DBS) Systems, can provide clinicians with simple and actionable data for efficient programming in the treatment of people living with Parkinson’s disease or essential tremor.
“The ability to see the precise placement of DBS Systems enables us to target therapy to meet individual needs,” Mustafa Saad Siddiqui, M.D., professor of Neurology and Neurosurgery at Wake Forest School of Medicine and medical director of the DBS program at Atrium Health Wake Forest Baptist. “The new features in the Vercise Neural Navigator 5 are expected to help further reduce the time needed to adjust stimulation and minimize potential side effects, allowing us to optimize treatment benefits for each patient.”
The Vercise Neural Navigator 5 Software, with STIMVIEW™ XT Technology, is the latest addition to the fully integrated portfolio of image guided programming solutions for Boston Scientific DBS Systems. Developed in collaboration with Brainlab AG, a leading software-driven medical technology company, these tools have demonstrated a reduction in programming time by 56%1 and provide real-time visualization and stimulation of each person’s unique brain anatomy.
The Vercise Neural Navigator 5 Software includes an enhanced user interface that displays patient data in a simplified format and gives clinicians access to advanced settings for increased therapy delivery. The software is designed to enable more flexibility to better manage the evolving needs of individual patients at any stage of their condition.
"Developing meaningful tools to help physicians provide personalized treatments for their patients delivers on our promise to advance our technologies for people living with neurological conditions,” said Jim Cassidy, president, Neuromodulation, Boston Scientific. “Providing effective DBS therapy is complex and can be time-consuming. This software will help streamline the process and allow for more doctor-patient interaction time."
For more information about the Vercise Genus DBS Systems and image guided programming software, please visit STIMVIEWXT.com.
About the Boston Scientific DBS System
The fourth-generation Vercise Genus™ Deep Brain Stimulation (DBS) System is designed to treat the symptoms of Parkinson’s disease and essential tremor by delivering targeted electrical stimulation via surgically-implanted leads in the brain connected to an implantable pulse generators (IPG). The portfolio, approved for conditional use in a magnetic resonance imaging (MRI) environment2, consists of a family of Bluetooth-enabled, rechargeable and non-rechargeable, IPGs that power the Vercise Cartesia™ Directional Leads, designed to provide optimal symptom relief.
About Boston Scientific
Boston Scientific transforms lives through innovative medical solutions that improve the health of patients around the world. As a global medical technology leader for more than 40 years, we advance science for life by providing a broad range of high performance solutions that address unmet patient needs and reduce the cost of healthcare. For more information, visit www.bostonscientific.com and connect on Twitter and Facebook.
Cautionary Statement Regarding Forward-Looking Statements
This press release contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934. Forward-looking statements may be identified by words like "anticipate," "expect," "project," "believe," "plan," "estimate," "intend" and similar words. These forward-looking statements are based on our beliefs, assumptions and estimates using information available to us at the time and are not intended to be guarantees of future events or performance. These forward-looking statements include, among other things, statements regarding clinical trials; our business plans and product performance and impact and new and anticipated product approvals and launches. If our underlying assumptions turn out to be incorrect, or if certain risks or uncertainties materialize, actual results could vary materially from the expectations and projections expressed or implied by our forward-looking statements. These factors, in some cases, have affected and in the future (together with other factors) could affect our ability to implement our business strategy and may cause actual results to differ materially from those contemplated by the statements expressed in this press release. As a result, readers are cautioned not to place undue reliance on any of our forward-looking statements.
Factors that may cause such differences include, among other things: future economic, competitive, reimbursement and regulatory conditions; new product introductions; demographic trends; intellectual property; litigation; financial market conditions; manufacturing, distribution and supply chain disruptions and cost increases; and future business decisions made by us and our competitors. All of these factors are difficult or impossible to predict accurately and many of them are beyond our control. For a further list and description of these and other important risks and uncertainties that may affect our future operations, see Part I, Item 1A – Risk Factors in our most recent Annual Report on Form 10-K filed with the Securities and Exchange Commission, which we may update in Part II, Item 1A – Risk Factors in Quarterly Reports on Form 10-Q we have filed or will file hereafter. We disclaim any intention or obligation to publicly update or revise any forward-looking statements to reflect any change in our expectations or in events, conditions or circumstances on which those expectations may be based, or that may affect the likelihood that actual results will differ from those contained in the forward-looking statements. This cautionary statement is applicable to all forward-looking statements contained in this document.
CONTACTS:
Lisa Ferrer
Media Relations
+1 (805) 561-4075 (mobile)
Lisa.Ferrer@bsci.com
Lauren Tengler
Investor Relations
+1 (508) 683-4479
BSXInvestorRelations@bsci.com
References:
- Lange, F et al. Reduced Programming Time and Strong Symptom Control Even in Chronic Course Through Imaging-Based DBS Programming, Front Neurol 2021 Nov 8;12:785529
- The Vercise Genus™ DBS System provides safe access to full-body 1.5T MRI scans when used with specific components and the patient is exposed to the MRI environment under specific conditions defined in the supplemental manual ImageReady™ MRI Guidelines for Boston Scientific DBS Systems.